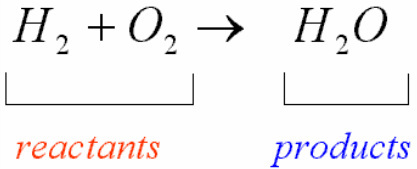|
In this page you will find a basic introduction to matter and terms describing matter and chemical reactions.
Word EquationsA chemical equation is broken into two parts by an arrow pointing to the right. The chemicals that are on the left are known as the reactants and are considered the ingredients or starting materials for the reaction. The arrow itself means "yields" or "reacts to form" and should be read this way. The chemicals on the right are known as the products and are the new materials that were made in the reaction.
Exothermic vs. Endothermic ReactionsA chemical reaction that releases energy is called an exothermic reaction. During these reactions the beaker (or test tube) where the reaction is taking place will feel hot because energy is being release from the reaction on to your hand.
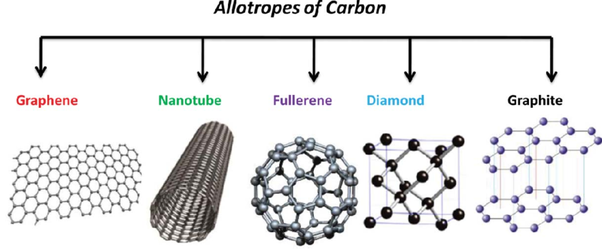
A chemical reaction that absorbs energy is called an endothermic reaction. During these reactions the beaker (or test tube) where the reaction is taking place will feel cold because the reaction is absorbing the energy from your hand as you touch it. AllotropesAtoms of the same element can be found in different forms. The picture below shows several different forms of the element carbon including: graphene, nanotube, fullerene, diamond, and graphite.
|
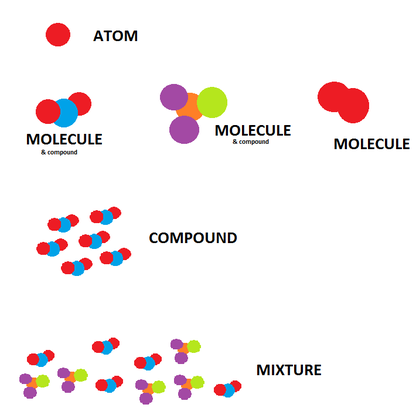
Atoms, Elements, and MoleculesAtoms are the smallest parts of matter that still retain the properties of that matter. Atoms combine to form molecules, and all matter is made of atoms and molecules.
|